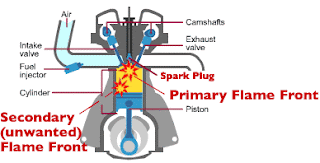
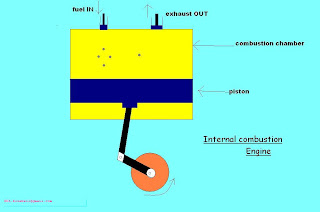
Knocking
A petrol/diseal engine basically works on the combustion of
oil in the presence of
Air which produces enough thrust by spark and burst to push
a piston
Forcibly.
The piston in turn rotates the gears of engine in circular
manner as shown.
In petrol engine
The saturated long chains of hydrocarbons (petrol oil) is
too good for combustion/burning.
It disturbs the harmony in which the piston moves up and
down.
This in turn produces crackling noise (just like rattling of
hammer).
This is called knocking of a petrol engine.
In diesel engine
In a similar but exactly opposite way knocking is observed in Diesel engine.
Diesel contains compounds which are highly branched or
aromatic which delays the combustion of the oil in the engine.
This delay disturbs the harmony in which the piston moves up
and down and causes knocking of diesel engine.
Disadvantages or drawbacks of knocking
* Causes harm to engine.
* Wastage of fuel as well energy produced by it.
* Harms piston and cylinder.
* Increases maintenance cost.
Measurement of knocking
In petrol engine
The knocking in petrol engine is measured by Octane number.
Octane number is the percentage of iso-octane present in
mixture of
iso-octane and n-heptane.
iso-octane is a compound which has very good, calm and
controlled combustion properties.
n-heptane detonates and burns readily before it is expected
to burn.
This causes knocking of petrol engine.
Thus, for Reference and convention
Octane number of iso-octane is taken as 100
Octane number of n-heptane is taken as 0
In diesel engine
Cetane number is used to measure knocking in diesel engine.
Cetane number is percentage of n-hexadecane present in
mixture of n-hexadecane
and methylnapthalene.
In order to appreciate the diesal engine one needs to know how it works.
Working of diesel engine
A diesel engine demands more arrangements as compared to its
peer petrol engine.
1. Firstly, air is pumped and compressed in the engine to attain
500C-700C temperatures.
2. Then oil is injected inside.
3. Atomization of oil begins at these elevated temperatures
and carbon of diesel combines with oxygen of air
to form CO2 (other
gases are also produced because of impurities).
4. The steps 2 and 3 have to be as fast as possible (1/500
th of a second).
The presence of aromatic and branches hydrocarbons produces
delayed combustion
And ignition lag causing Knocking of engine.
So, for reference and convection
n-hexadecane ignites readily
Thus,
it has Cetane number 100
whrereas methylnapthalene delays combustion
Thus, it has Cetane number 0